Avedro KXL® System
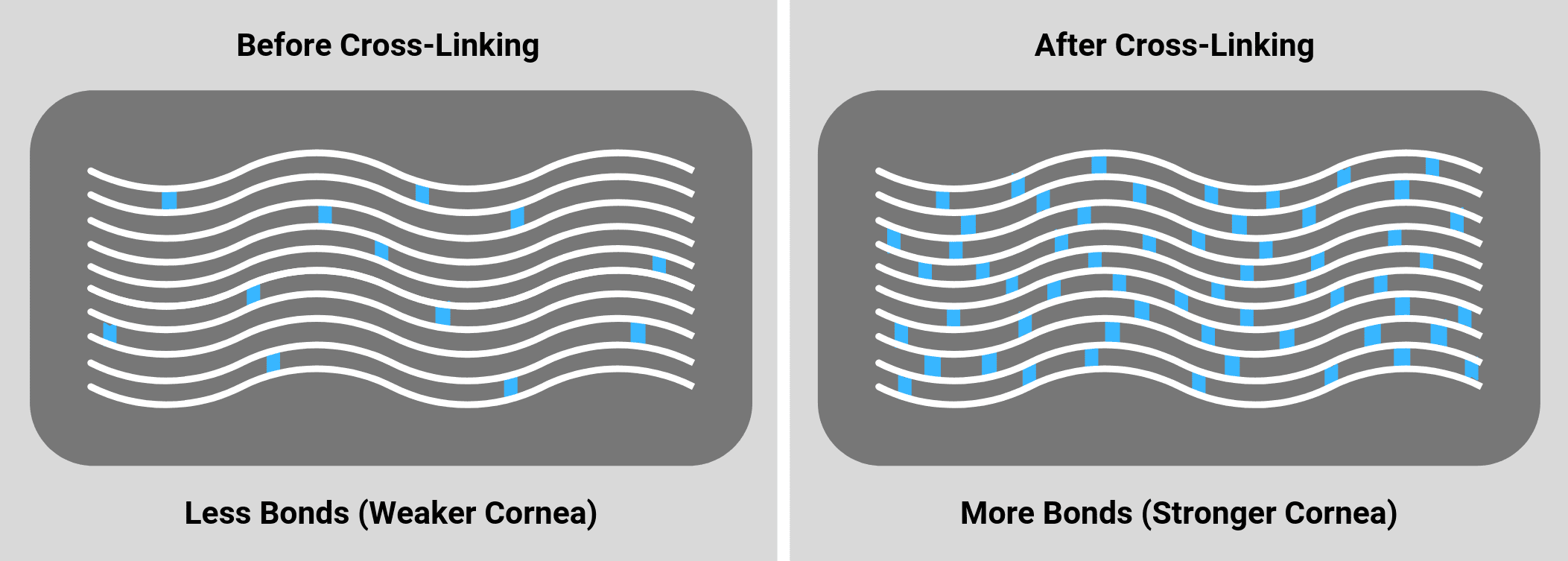
Corneal cross-linking technology has been available in Europe for over a decade. Currently in the US, the only FDA approved cross-linking platform is the Avedro KXL® System, which utilizes the Photrexa® and Photrexa® Viscous riboflavin formulations. Many insurance providers are beginning to recognize the medical necessity of this procedure. In fact, there are a large list of providers that have already implemented policies that provide coverage towards this FDA approved corneal cross-linking system.
Cross-Linking Candidates
During an initial evaluation, we will measure the thickness and shape of your cornea to determine if you are a good candidate for a cross-linking procedure. We will use a corneal topographer to provide a detailed view of your corneal surface. Cross-linking works best when treating an early stage of Keratoconus. The treatment is not designed to reverse the corneal condition but prevent it from getting worse. This is an outpatient procedure that takes no more than 60-90 minutes to complete. For further inquiries, please contact our team to schedule an appointment for a cross-linking consultation.
At Florida Eye Specialists & Cataract Institute, we remain on the forefront of surgical innovation. Call today or request an appointment to meet with our world-class corneal specialists.